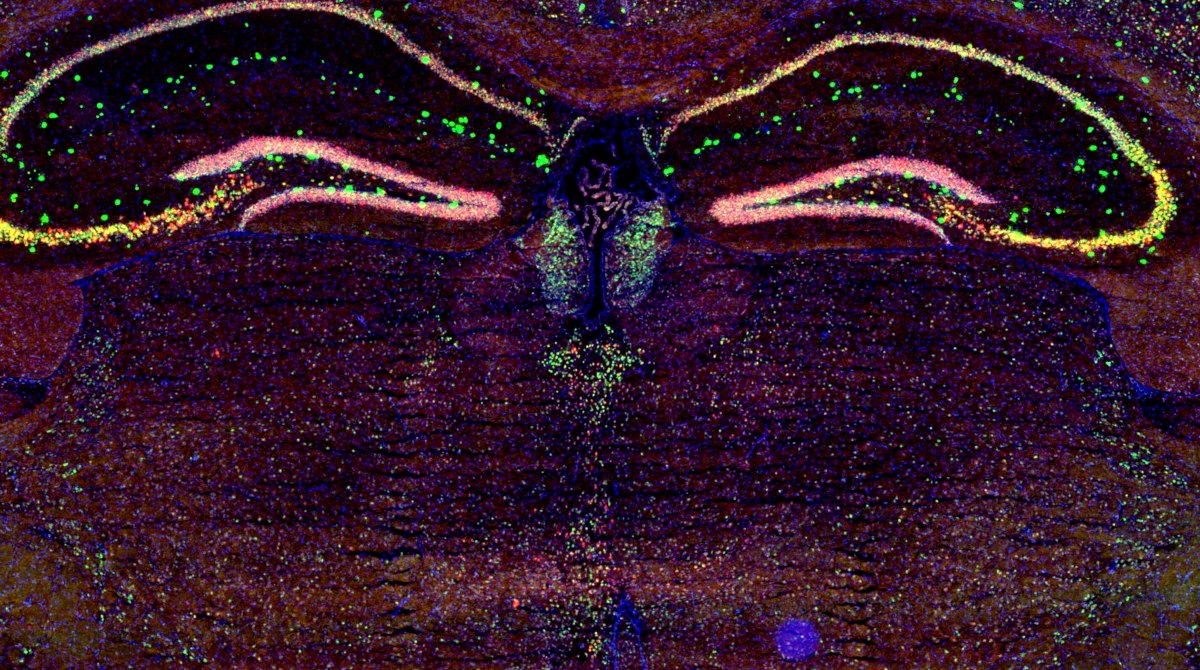
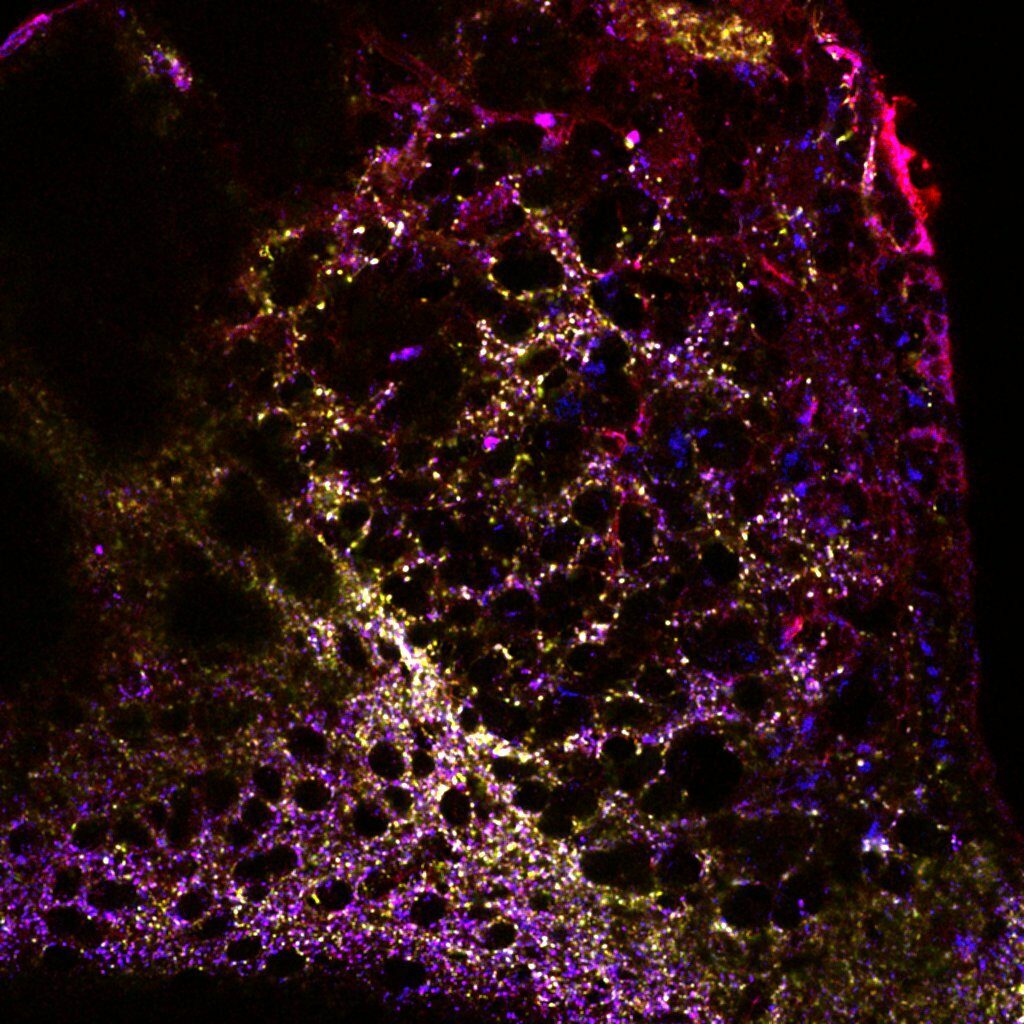
Our scientific interests are focused on how molecular mechanisms contributing to the processing of inhibitory and excitatory inputs in neurons are integrated into neural circuits and determine affective, emotional behaviors.
In the laboratory, in particular, we concentrate on the intralaminar and midline thalamus (IL/MDLs), associative higher-order nuclei controlling emotional and attentional states and wake/sleep transitions, and on the epithalamic Medial Habenula (MHb), which controls aversion. The thalamus and the epithalamus are key hubs for the transfer of information between the anterior and the posterior brain. Although thalamus and epithalamus show a largely heterogeneous connectivity pattern, their circuital organization in rodents is simple and shows common features. Namely, no local circuitry is present in the vast majority of both thalamic and epithalamic subregions. Here, nuclei are indeed mostly composed of relay neurons that receive the incoming information and retransmit it without forming local collaterals. Information is thus processed at individual neuronal level by how cell firing patterns are affected by the incoming activity and by the activation of local molecular events. In this context, both the IL/MDLs and the MHb, and up/downstream areas, are endowed with unique (non)synaptic mechanisms contributing to information treatment.
For our research, we use a bottom-up approach. Starting from the analysis of both the morphological and the physiological organization and ex vivo properties of synaptic connections, our approaches extend to a comprehensive investigation of the in vivo behavioral outcomes elicited by interfering with identified molecular actors. To do this we apply state-of-the-art optogenetic and chemogenetic viral approaches in transgenic mouse models, in combination with more conventional morphological tools.
In recent years, our team provided important contributions in the domains of thalamic and medial habenular physiology. As an example, in the MHb our work led to the first demonstration of the existence of novel excitatory glycinergic NMDA receptors containing the unconventional GluN3A subunit. Our finding that these receptors are mandatory for the emergence of conditioned aversive states in adult mice (Otsu et al., Science, 2019) extended the spectrum of physiological functions associated with the typically inhibitory neurotransmitter glycine.
If you want to explore the possibility of joining us either as a post-doctoral fellow or for a PhD program, please contact Marco Diana or Eric Schwartz.
Team leader
- Marco Diana, Senior Research Scientist, PI, CNRS, Team 6 - Synapses of Affective Behaviors, +33 1 76 53 43 41 (switchboard)
Members
- Eléonore Hardy, Postdoctoral Researcher, Team 6 - Synapses of Affective Behaviors, +33 1 76 53 43 41 (switchboard)
- Dersu Özdemir, Postdoctoral Researcher, Team 6 - Synapses of Affective Behaviors, +33 1 76 53 43 41 (switchboard)
- Eric Schwartz, Assistant Professor, Team 6 - Synapses of Affective Behaviors, +33 1 76 53 43 41 (switchboard)
Recent Publications
Articles
- Model of negative affect induced by withdrawal from acute and chronic morphine administration in male mice.
Ozdemir D, Meyer J, Kieffer BL, Darcq E : Sci Rep, 2024 - Rebound activation of 5-HT neurons following SSRI discontinuation.
Collins HM, Gullino LS, Ozdemir D, Lazarenco C, Sudarikova Y, Daly E, Pilar Cuéllar F, Pinacho R, Bannerman DM, Sharp T : Neuropsychopharmacology, 2024 - Advances in the characterization of negative affect caused by acute and protracted opioid withdrawal using animal models.
Ozdemir D, Allain F, Kieffer BL, Darcq E : Neuropharmacology, 2023 - Local glycolysis fuels actomyosin contraction during axonal retraction.
Santos R, Lokmane L, Ozdemir D, Traoré C, Agesilas A, Hakibilen C, Lenkei Z, Zala D : J Cell Biol, 2023 - Effect of selective serotonin reuptake inhibitor discontinuation on anxiety-like behaviours in mice.
Collins HM, Pinacho R, Ozdemir D, Bannerman DM, Sharp T : J Psychopharmacol, 2022 - Habenular Neurons Expressing Mu Opioid Receptors Promote Negative Affect in a Projection-Specific Manner.
Bailly J, Allain F, Schwartz E, Tirel C, Dupuy C, Petit F, Diana MA, Darcq E, Kieffer BL : Biol Psychiatry, 2022 - Astroglial Cx30 differentially impacts synaptic activity from hippocampal principal cells and interneurons.
Hardy E, Cohen-Salmon M, Rouach N, Rancillac A : Glia, 2021 - Adult medial habenula neurons require GDNF receptor GFRα1 for synaptic stability and function.
Fernández-Suárez D, Krapacher FA, Pietrajtis K, Andersson A, Kisiswa L, Carrier-Ruiz A, Diana MA, Ibáñez CF : PLoS Biol, 2021